Polytetrafluoroethylene, PTFE, has a remarkably low dielectric constant (approx 2.1) and an incredibly low dissipation factor (dielectric loss).
These properties qualify PTFE films as high-value materials for high-performance insulation in wiring and cables, high-frequency printed circuit boards, particularly in telecommunications and aerospace applications.
But what is behind PTFE's dielectric strength?
The Chemical Structure Behind Dielectric Strength
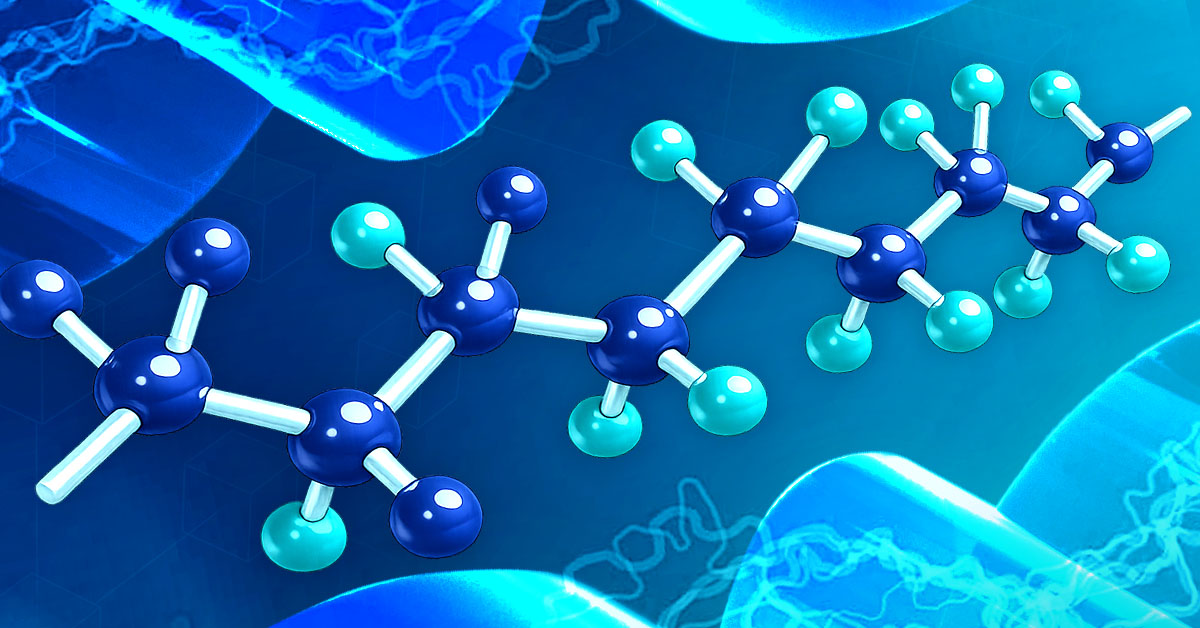
PTFE’s dielectric properties primarily derived from its unique carbon–fluorine molecular structure: an internal carbon backbone completely shielded by a dense, spiraling sheath of fluorine atoms.
This structure creates an impenetrable "molecular armor", completely blocking corrosive chemicals from reaching the carbon spine.
Because the structure is so stable and non-reactive, there are virtually no free electrons or mobile ions to carry an electric current, resulting in its exceptionally high volume resistivity - i.e. the ability to oppose the flow of electric current through its bulk - and excellent insulation resistance.
PTFE repeat unit:
[−CF2−CF2−]n

In Details:
- Electronic Polarizability and The C–F Bond: The C–F bond is extremely strong and highly stable.
Fluorine is very electronegative, so it tightly holds electron density and makes the bond difficult to polarize or break. Because the electrons are not easily displaced by an electric field, PTFE has low electronic polarizability, which contributes to its low dielectric constant and low dielectric loss. - Molecular Symmetry and Dipole Cancellation
Although each C–F bond is polar, the dipoles around the carbon backbone largely cancel each other. That means PTFE has very little permanent dipole response.
Materials with strong mobile dipoles tend to absorb electromagnetic energy and show higher dielectric loss; PTFE does not. - PTFE contains no oxygen-, nitrogen-, or sulfur-containing functional groups such as hydroxyl, carbonyl, amide, or ester groups. Those groups often increase polarity, moisture uptake, and dielectric loss. PTFE is almost entirely a saturated fluorocarbon, so it is chemically inert, hydrophobic, and electrically stable.
At Versiv, we have over 40 years of experience with high-performance fluoropolymer composites.
Contact Us today!